 The value of n will be always be a whole number that represents how far away the electron is from the nucleus. The symbol used to describe the first quantum number is n. The first quantum number, also called the principal quantum number, determines the energy level or shell that the electron is in. This may get a little wordy, but trust us. Each electron in an atom has his own distinct set of four quantum numbers just like your phone number is different from everyone else's. Think of it as an electron's cell phone number. These are the numbers assigned to each particular electron in an atom. To begin to understand how electrons really behave, we need to learn about quantum numbers. Unlike the old planetary model of the atom, this new quantum-mechanic model describes a less certain electron orientation. Let's think of electron movement in terms of probability, or where the electrons are most likely to be found-it's like finding the odds of rolling a particular number with two dice. That is an old-fashioned idea we should throw out the window. In other words, electrons are not like roller coasters moving along a stationary track. The old way of thinking about electron movement in fixed orbits around the nucleus is incorrect. Electrons, however, have their own way of 'dancing' within the cloud. Think of it as one big electron disco where all of the outer electrons are moving about and shaking their groove things. The electron cloud we're talking about is the place where electrons move around outside of the nucleus. 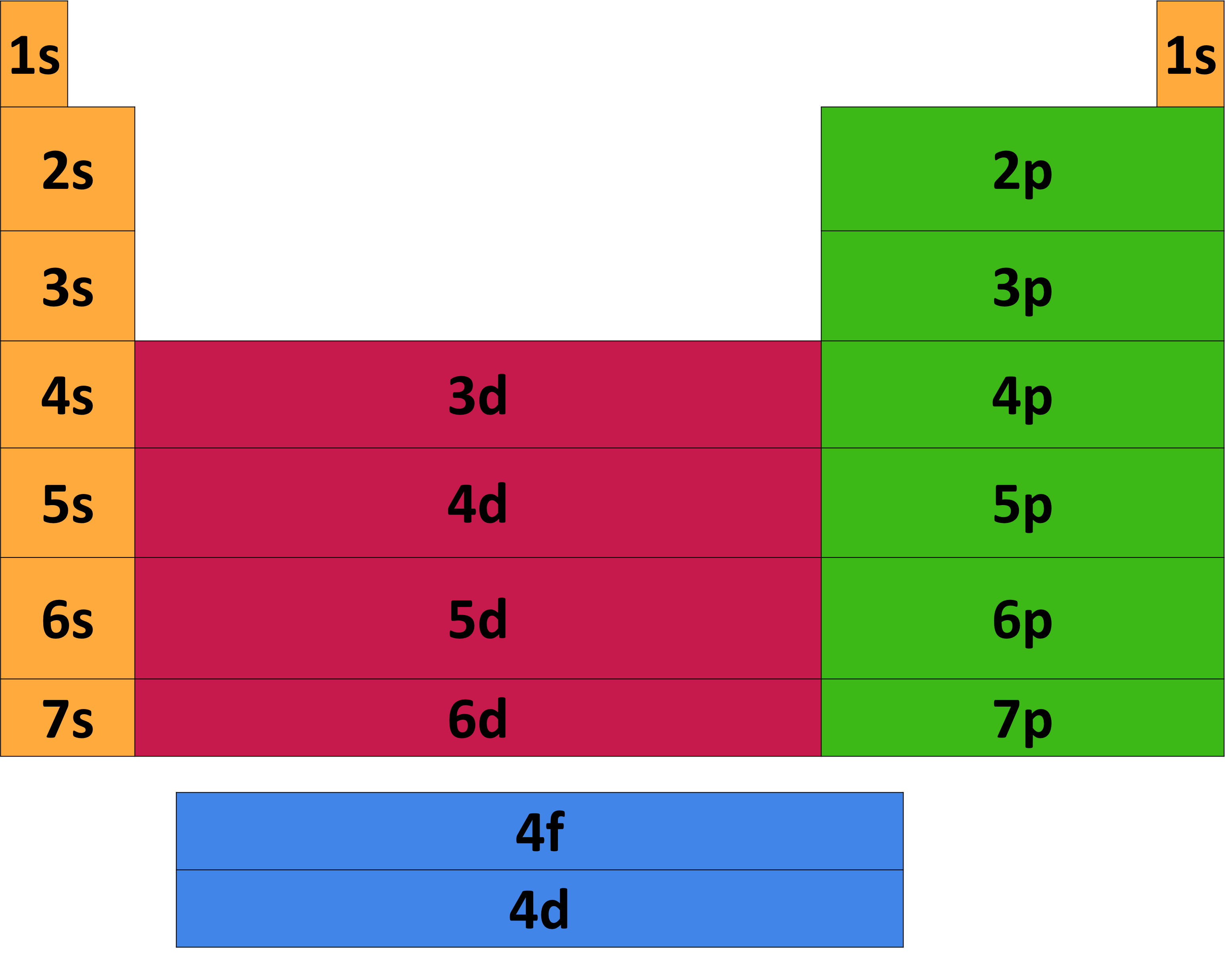 
This cloud is different from the electron "pool" which is reserved solely for metallic crystals and metallic bonding. That's why it's important to understand exactly what's going on in the electron cloud. This need is what drives atoms to bond with each other. Atomic Orbitals and Electron Configurations All Types of Bonds and OrbitalsĪn atom needs to have 8 electrons in its outer shell in order to feel complete. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
